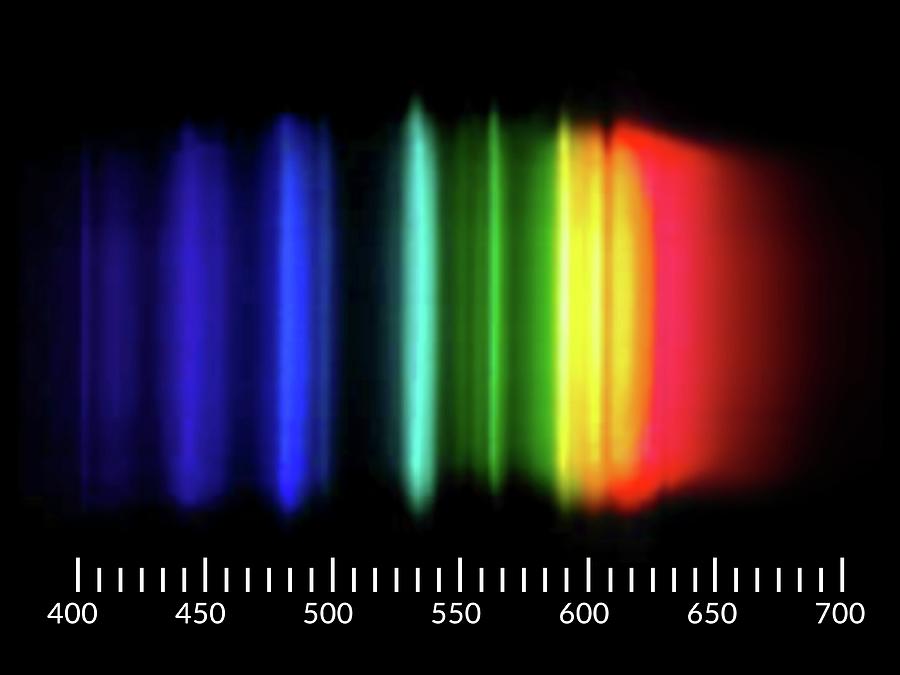 
SBFI is 18-fold more selective for Na + than for K +. Emission hand forms of several elements obtained with a photon-counting spectrometer are compared with previously published spectra. There are two other blue lines at 404.656 nm and 407.781 nm and a weak line at 491.604 nm. The dissociation constant (K d) of SBFI for Na + is 3.8 mM in the absence of K +, and 11.3 mM in solutions with a combined Na + and K + concentration of 135 mM, which approximates physiological ionic strength. The prominent mercury lines are at 435.835 nm (blue), 546.074 nm (green), and a pair at 576.959 nm and 579.065 nm (yellow-orange). The sodium D-line is actually a pair of closely spaced spectroscopic lines seen in the emission spectrum of sodium atoms. The inset shows that there are two closely spaced emission lines with wavelengths of 589.0 and 589.6 nm. Figure 8.1.3: Atomic emission spectrum for Na at a temperature of 3000 K. At the right of the image are the spectral lines through a 600 line/mm diffraction grating. Analyzing The Emission Spectra of Sodium Symbol: Na Atomic number: 11 Flame color: Yellow Orange (visible region) Wavelength of light emitted: 600 nm. As seen in Figure 8.1.3, the emission spectrum for Na is dominated by the pair of lines with wavelengths of 589.0 and 589.6 nm. In aerosol form, sodium is applied to nuclear, electronic industries, and medical fields. Sodium Green (sodium bound) is a fluorescent compound with an excitation peak at 507 nm and an emission peak at 532 nm. Figure 10. 
Such signs are excited by voltages of a few thousand volts produced by a transformer that raises the voltage of the ordinary AC line voltage.Īt left is a mercury spectral tube excited by means of a 5000 volt transformer. Introduction Sodium is one of the most useful ingredients in everyday life. Atomic emission occurs when a valence electron in a higher energy atomic orbital returns to a lower energy atomic orbital. This is a section of the sign shown below, which has a central neon section and another gas mixture producing blue light around it. Then the image below was reduced and superimposed on the image above, because with the exposure reasonable for the bright tube, only the red lines were visible on the photograph. The sodium spectrum is dominated by the bright doublet known as the Sodium D- lines at 588.9950 and 589.5924 nanometers. The image below is composed of segments of three photographs to make the yellow and green lines more visible along with the much brighter red lines. This is an attempt to give a reasonable accurate picture of the appearance of the neon spectrum, but both the images are composite images.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
